Proliferation of a rare neuroreceptor may underlie the intensification of craving that cocaine abusers experience during their first weeks of abstinence. NIDA-funded researchers found that, in rats, the quantity of these receptors in the nucleus accumbens (NAc) increased from the first to the 45th drug-free day and correlated with an increase in the animals' drug-seeking.
The implicated receptors are a subtype of the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor. AMPA receptors regulate cellular responses to the neurotransmitter glutamate throughout the brain, including in the NAc, an area that shapes motivation by linking experience to feelings of reward and satisfaction. Most AMPA receptors include subunits called GluR1 and GluR2, but those newly generated following cocaine withdrawal lack GluR2.
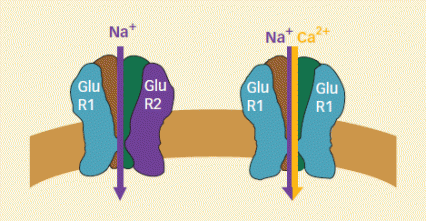 New Receptors Appear During Withdrawal From Cocaine: AMPA receptors respond to glutamate signals within the brain and play a role in cocaine seeking. These receptors have four subunits that typically include one called GluR2 (left). However, during withdrawal from cocaine, new AMPA receptors that lack GluR2 (right) appear in the membranes of nerve cells in the brain's nucleus accumbens. The new receptors are more permeable to calcium and have a higher ion flow when activated than the more common AMPA receptors.
New Receptors Appear During Withdrawal From Cocaine: AMPA receptors respond to glutamate signals within the brain and play a role in cocaine seeking. These receptors have four subunits that typically include one called GluR2 (left). However, during withdrawal from cocaine, new AMPA receptors that lack GluR2 (right) appear in the membranes of nerve cells in the brain's nucleus accumbens. The new receptors are more permeable to calcium and have a higher ion flow when activated than the more common AMPA receptors."Switches in the subunit composition of AMPA receptors have been uncovered in other neural modifications, but our study describes the most long-lasting change that has yet been found in any brain region," says Dr. Marina Wolf of the Chicago Medical School at Rosalind Franklin University of Medicine and Science. According to Dr. Wolf, her team's results are particularly exciting because they show how long-lasting modifications can be produced in the adult brain and because the brain alteration is directly linked to a disease-related behavioral change.
Nose Pokes and Receptors
Scientists have long recognized that cocaine craving intensifies, or incubates, for several weeks following withdrawal from the drug. Cocaine abusers' reports of craving and their risk of relapse both peak after several weeks of abstinence.
Prior NIDA-funded research has shown that drug craving involves a complex neuronal circuit that includes cortical and limbic regions, as well as NAc. However, there is evidence that glutamate inputs into the NAc are part of a final common pathway for drug seeking. What's more, AMPA-type glutamate receptors are increasingly recognized as key players in the rewiring of brain circuits in response to many kinds of experience. Dr. Wolf's team therefore focused on testing the role of NAc AMPA receptors in the incubation of cocaine craving.
The team's first step was to induce craving incubation in rats. Animals were trained to poke their noses into a hole in a test chamber to elicit an intravenous infusion of cocaine. Each infusion was paired with a light or light-plus-tone cue, enabling the rats to learn to associate these cues with cocaine availability. Rats had access to cocaine for 6 hours a day, enabling a high level of drug intake. After 10 days of this regimen, the researchers initiated cocaine withdrawal by halting the rats' access to the drug.
At intervals during the rats' withdrawal from the drug, the researchers returned them to the test chamber. The rats resumed poking their noses in the hole, but the poking now brought no cocaine. The researchers counted the number of nose pokes as a measure of the animals' desire for the drug and the willingness to expend effort to obtain it. The rats poked more than twice as much on withdrawal day 45 than on withdrawal day 1, demonstrating that their urge to obtain cocaine had increased—incubated—during the interval.
Biochemical assays of the rats' brain tissue showed that the number of GluR1 receptor subunits in the NAc doubled between withdrawal day 1 and day 45. There was no concomitant increase in GluR2 subunits, however, indicating that the NAc cells had generated a new complement of GluR2-lacking receptors during this interval. No other brain area exhibited AMPA changes, nor were there changes in another common type of glutamate receptor, the NMDA receptor.
The researchers next established a causative role for the newly formed GluR2-lacking AMPA glutamate receptors in the rats' drug-seeking behavior. They injected a chemical (1-naphthyl acetyl spermine, or NASPM) that prevents glutamate from binding to GluR2-lacking AMPA glutamate receptors into the NAc of cocaine-exposed rats and showed that, in this situation, incubation is inhibited. Derived from a spider toxin, the chemical reduced cocaine-seeking behavior on withdrawal day 45 by more than half.
Mechanisms of Incubation
The new findings may be explained in terms of previous research that has shown that what happens when glutamate binds to an AMPA receptor depends, in part, on whether the receptor contains GluR2. All AMPA receptors contain an ion channel that allows sodium entry, which excites neurons by depolarizing them. AMPA receptors that lack GluR2 admit calcium as well as sodium. This leads to a stronger depolarization as well as other consequences for intracellular signaling.
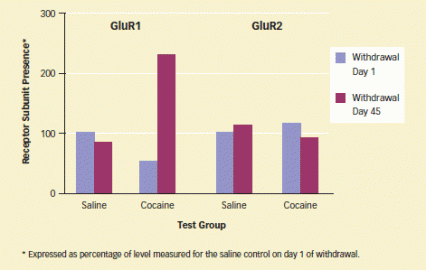 AMPA Receptors Produced During Cocaine Withdrawal Lack GluR2 Subunits: Between day 1 and day 45 of cocaine withdrawal, the number of GluR1 receptor subunits increased dramatically, while the number of GluR2 subunits stayed about the same.
AMPA Receptors Produced During Cocaine Withdrawal Lack GluR2 Subunits: Between day 1 and day 45 of cocaine withdrawal, the number of GluR1 receptor subunits increased dramatically, while the number of GluR2 subunits stayed about the same."Calcium entry through the GluR2-lacking AMPA receptor channel may contribute to cocaine-induced brain changes by activating signaling pathways that are not in play when other types of AMPA receptors are stimulated in normal rats," Dr. Wolf suggests. For example, calcium influx into brain cells via glutamate receptors has been shown to promote lasting sensitization of the cell to future glutamate signals in a process, called long-term potentiation, that encodes information and experience during learning. Such sensitization in the NAc, with its role in motivation and reward, would be compatible with incubation of cocaine craving and seeking.
Why should cocaine withdrawal precipitate AMPA receptor proliferation in the NAc? Dr. Wolf and colleagues note that during cocaine withdrawal, areas of the brain that release glutamate into the NAc are hypoactive. Possibly, the NAc ramps up its AMPA receptor complement to compensate for the resulting reduction in incoming neurotransmitter.
"This compensatory change backfires when the animal encounters a cocaine-related cue and glutamate is released into the NAc," says Dr. Wolf. "This occurs because the responsiveness of the NAc neurons to glutamate has been increased due to synaptic incorporation of GluR2-lacking AMPA receptors. We propose that this enhanced responsiveness of NAc neurons underlies increased cue-induced cocaine craving."
Clinical Applications
In recovering addicts, exposure to environmental cues previously associated with drug use, such as cocaine paraphernalia, is an important trigger for cocaine craving leading to relapse. Dr. Wolf's work on cue-induced cocaine craving in rats suggests that GluR2-lacking AMPA glutamate receptors might be a useful target for medications designed to reduce the frequency of relapse. While blocking glutamate receptors may lead to serious side effects, selective blockade of GluR2-lacking AMPA receptors might be less problematic because these receptors are a minority population in the normal brain, according to Dr. Wolf.
NASPM cannot be used in people because it works only when injected directly into the brain. Scientists may, however, develop similar blockers in the future that could be taken as pills or by injection. Such a medication might be used to dampen cue-induced cocaine craving during the first few months of withdrawal, when abstinent cocaine addicts are particularly prone to cue-induced relapse, Dr. Wolf says.
Dr. Jerry Frankenheim of NIDA's Functional Neuroscience Research Branch agrees that a drug that acts like NASPM could potentially be a useful treatment for blocking cue-induced cocaine seeking. "NIDA's medications development group is also looking into NASPM as a lead towards a therapy to prevent relapse," he says.
He adds that NASPM-like compounds may be useful for other cue-induced drug cravings as well as in treating brain disorders, including neuronal cell death and epilepsy.
"The expression of GluR2 in neurons is altered not only by drugs of abuse but also, in certain vulnerable neurons, after seizures, ischemia, or administration of some other drugs. For example, in some neurons, ischemia triggers down-regulation of GluR2 and enhances AMPA receptor-mediated calcium and zinc influx that may mediate the death of these neurons. Therefore, NASPM-like medications may obtain several therapeutic applications," Dr. Frankenheim says.
"Dr. Wolf's work represents part of an exemplary, synergistic collaboration with members of NIDA's Intramural Research Program, including Dr. Yavin Shaham, who in 2001 first reported incubation of cocaine craving in an animal model," says Dr. Frankenheim.
Source
Conrad, K.L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454(7200):118-121, 2008. [Abstract]
