Results from NIDA’s Clinical Trials Network (CTN) affirm Centers for Disease Control and Prevention (CDC) recommendation that health care facilities serving high-risk populations offer onsite HIV testing. Patients who were offered testing in the centers where they received treatment for substance abuse were four times as likely to be tested as patients who were referred offsite.
 HIV Rapid Testing and Risk-Reduction Counseling Outcomes Patients were more likely to take a rapid HIV test when substance abuse treatment programs offered the test onsite rather than referred for offsite testing. Patients were roughly as likely to accept and learn their HIV status whether the offer of onsite testing was accompanied by 30 minutes of risk reduction counseling or by 5 minutes of brief information on the testing procedure.
HIV Rapid Testing and Risk-Reduction Counseling Outcomes Patients were more likely to take a rapid HIV test when substance abuse treatment programs offered the test onsite rather than referred for offsite testing. Patients were roughly as likely to accept and learn their HIV status whether the offer of onsite testing was accompanied by 30 minutes of risk reduction counseling or by 5 minutes of brief information on the testing procedure.
- Text description
-
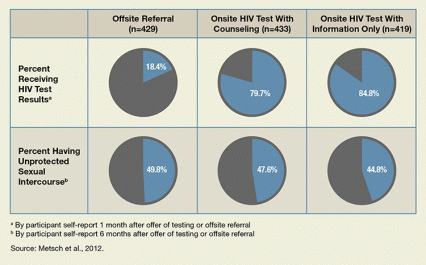
Six pie charts showing the percentage of patients who received HIV test results and the percentage of patients who reported having unprotected intercourse.
Percent Receiving HIV Test Results: Offsite Referral 18.4%; Onsite HIV Test With Counseling 79.7%; Onsite HIV Test with Information Only 84.8% (by participant self-report 6 months after offer of testing or offsite referral).
Percent Having Unprotected Sexual Intercourse: Offsite Referral 49.8%; Onsite HIV Test With Counseling 47.6%; Onsite HIV Test with Information Only 44.8% (by participant self-report 6 months after offer of testing or offsite referral).
The recent trial also supported current CDC advice to omit pretest risk reduction counseling. Study participants whose test offer was prefaced with 5 minutes of information about the procedure, and those who received 30 minutes of risk reduction counseling, accepted testing at roughly equal rates. Both groups also reported similar frequencies of sexual risk behaviors during the 6 months subsequent to their test offers (see figure).
Twelve CTN-affiliated treatment centers throughout the United States took part in the trial, which was co-led by Dr. Lisa Metsch, of Columbia University’s Mailman School of Public Health and the University of Miami Miller School of Medicine, and Dr. Grant Colfax, of the San Francisco Department of Public Health. (Dr. Colfax is now Director of the White House Office of National AIDS Policy.) Collectively, the centers provide inpatient, outpatient, and opioid replacement therapies.
Staff in each center offered HIV testing or referral for testing to all adult patients who said they had not been tested within the past 12 months and either believed themselves to be HIV-negative or didn’t know their HIV status. A total of 1,281 participants were randomized in the trial; at each center, roughly one-third of patients were randomly selected for referral for offsite testing, and the remaining two-thirds to receive an offer of onsite testing accompanied by either:
- Risk reduction counseling: Staff explained the routes of HIV transmission, provided a personalized risk assessment, and motivated participants to get tested.
or; - Brief procedural information: Staff described the rapid HIV test procedure, explained the test’s reliability, and told participants how long test results would take.
Interviews with participants 1 month after their test offers revealed that many more of those offered onsite testing learned their HIV status, compared to those referred offsite. Patients got tested and received their results at rates of: 79 percent with counseling and onsite testing; 84 percent with brief procedural information and onsite testing; and 18 percent with offsite referral.
Interviews with participants 6 months after their test offers found similar rates of unprotected vaginal and anal sex across all the groups. However, the availability of onsite testing was associated with reduced sexual risk behaviors among the 655 of these participants who had reported engaging in them at the start of the study. These patients reported the behaviors at rates of: 53 percent with pretest counseling and an onsite testing offer; 52 percent with brief procedural information and an onsite testing offer; and 61 percent among patients referred offsite.
The researchers concluded that brief procedural information with an offer of onsite testing was the intervention of choice. With less expenditure of staff time and resources, it yielded rates of test completion and sexual risk reduction that were equal to those obtained with counseling and an offer of onsite testing, and greater than those obtained with offsite referral. Dr. Bruce Schackman, of Weill Cornell Medical College in New York City, with Drs. Metsch and Colfax, and colleagues, also estimated that onsite testing without counseling resulted in a beneficial cost-effectiveness ratio for the CTN study population.
In this analysis, the research team used data from the trial, including patient demographics, prior testing history, test acceptance and receipt of results, undiagnosed HIV prevalence, and program costs. They used the Cost Effectiveness of Preventing AIDS Complications computer simulation model to project life expectancy, lifetime costs, and quality-adjusted life years (QALYs) for the HIV-infected individuals. The researchers calculated the cost per QALY with and without onsite testing, also taking into account the costs associated with testing individuals who turn out not to be HIV infected.
The results indicated that brief procedural information with an offer of onsite testing would extend life expectancy for HIV-infected individuals by an average of 3.7 years per person compared to no offer of testing or referral. Taking into account the cost of testing and the medical costs associated with longer survival, study results showed a cost-effectiveness ratio of $60,300 per QALY, well within the $100,000 per QALY figure that is sometimes used as the criterion for an acceptable level of cost effectiveness in the United States.
This study was supported by NIH grants: HHSN271200522071C, HHSN271200522081C, DA013038, DA013720, DA013732, DA015833, DA020036, DA027379, DA13034, DA13036, DA13720-09S, DA13727, DA15815, DA015612, AI042006, DA019809, DA028973, DA013727, DA13034, TR000128, DA015612, MH065869, DA013034, and DA015815.
Sources:
Metsch, L., et al. Implementing rapid HIV testing with or without risk-reduction counseling in drug treatment centers: results of a randomized trial. American Journal of Public Health 102(6): 1160-1167, 2012. Abstract
Schackman, B.R., et al. The cost-effectiveness of rapid HIV testing in substance abuse treatment: Results of a randomized trial. Drug and Alcohol Dependence 128(1-2):90-77, 2013. Abstract
