Men benefit more than women from nicotine replacement therapy (NRT) for smoking cessation because nicotine affects a key neuroreceptor differently in the two sexes, a NIDA-sponsored study suggests. The findings highlight the need for alternative therapies for women smokers, and point to the female hormone progesterone as a potential therapeutic target.
Dr. Kelly Cosgrove and colleagues at Yale University School of Medicine found that newly abstinent male smokers had more available units of a key nicotinic acetylcholine receptor (nAChR) than male nonsmokers. Among women, this was not the case: smokers had similar numbers of receptors compared to nonsmokers. The researchers hypothesize that in men, the brain adapts to the chronic presence of nicotine by adding receptors, but then needs nicotine to continue to function normally. Among women, a different adaptation takes place, which may increase susceptibility to sensory cues to smoke, and may be modulated by progesterone.
Smoking Patterns and Receptors
Along with the nicotine’s addictive effects, smoking provides people with other conditioned stimuli that reinforce the habit. Studies show that men are more sensitive than women to nicotine’s pharmacological effects, and that women are more susceptible than men to sensory and environmental cues to smoke.
“Men smoke more for nicotine reinforcement,” says Dr. Cosgrove, “while women smoke more for affect and stress regulation.” These patterns are paralleled in the two sexes’ contrasting responses to therapy: Men get more relief of craving from NRT, while women are helped more by devices that mimic the sensory concomitants of smoking, such as nicotine-free inhalers.
Dr. Cosgrove and colleagues hypothesized that the sexes’ divergent smoking patterns and responses to therapy arose from differences in smoking’s impact on nAChRs. The Yale researchers focused specifically on the subset of nAChRs that incorporate the β2 subunit in their structure (β2*-nAChRs). Past research has shown that nicotine binding to this subset, one of the most widespread in the brain, mediates the drug’s rewarding and other effects.
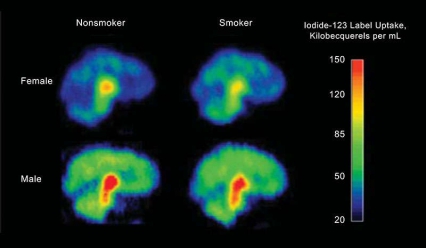 Figure 1. Single-Photon Emission Computed Tomography Imaging Reveals Smoking-Related Alterations in Receptor Availability Higher levels of radioactivity (measured in kilobecquerels per mL of brain tissue) indicate greater β2*-nicotinic acetylcholine receptor availability in these single-photon emission computed tomography (SPECT) images. To create the images, a newly abstinent smoker and a nonsmoker of each gender were injected with a radiolabeled tracer that binds to available receptors.
Figure 1. Single-Photon Emission Computed Tomography Imaging Reveals Smoking-Related Alterations in Receptor Availability Higher levels of radioactivity (measured in kilobecquerels per mL of brain tissue) indicate greater β2*-nicotinic acetylcholine receptor availability in these single-photon emission computed tomography (SPECT) images. To create the images, a newly abstinent smoker and a nonsmoker of each gender were injected with a radiolabeled tracer that binds to available receptors.In the study, male and female smokers underwent SPECT brain imaging (see Figure 1) after 7 to 9 days of abstinence, by which time most nicotine and nicotine metabolites would have cleared from their brains. The researchers compared the resulting images with SPECT images of nonsmokers. The comparison revealed that 26 male smokers had significantly higher β2*-nAChR availability than 26 male nonsmokers in the cortex (13 percent to 17 percent), striatum (16 percent), and cerebellum (17 percent), but similar availability in all other areas. Among females, 28 smokers had lower β2*-nAChR availability (-17 percent) than 30 nonsmokers in the thalamus, but similar availability elsewhere (see Figure 2).
Dr. Cosgrove says that the functions of the regions in which men and women incurred nicotine-induced β2*-nAChR changes fit the differences in their smoking patterns. The striatum, where men exhibited β2*-nAChR changes, generates feelings of pleasure and reward and regulates cognition and movement. The thalamus, where women showed β2*-nAChR changes, regulates the flow of sensory information between the cortex and areas of the brain associated with emotion and motivation.
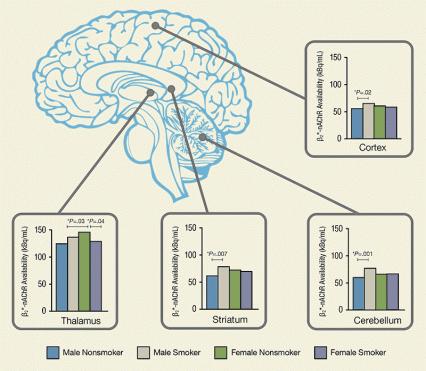 Figure 2. Male and Female Smokers Show Differences in Availability of Nicotine-Binding Receptor Male smokers who had been abstinent for 7 to 9 days had greater receptor β2*-nicotinic acetylcholine receptor (β2*-nAChR) availability than male nonsmokers throughout the cortex, cerebellum, and striatum, but not the thalamus. Female smokers had lower receptor availability than female nonsmokers in the thalamus. For measurement technique and units, see Figure 1.
Figure 2. Male and Female Smokers Show Differences in Availability of Nicotine-Binding Receptor Male smokers who had been abstinent for 7 to 9 days had greater receptor β2*-nicotinic acetylcholine receptor (β2*-nAChR) availability than male nonsmokers throughout the cortex, cerebellum, and striatum, but not the thalamus. Female smokers had lower receptor availability than female nonsmokers in the thalamus. For measurement technique and units, see Figure 1.Men’s Need for Nicotine
Male ex-smokers’ nicotine craving and the relief they receive from NRT may reflect their amplified β2*-nAChR availability, Dr. Cosgrove says. In this scenario, abstinent men’s expanded β2*-nAChR availability is the product of an adaptation that helps them tolerate chronic nicotine exposure, but becomes a liability once nicotine is withdrawn (see Figure 3).
In nonsmokers and smokers alike, the neurotransmitter acetylcholine binds to β2*-nAChRs (and other nAChRs) on the neuron membrane. Acetylcholine binding to nAChRs activates the neuron. Maintaining levels of neuronal activity that support normal function requires that there be sufficient receptors available for acetylcholine binding, but not too many.
In smokers, nicotine also binds to nAChRs. In the picture that is emerging from the Yale researchers’ and others’ work, nicotine binding to β2*-nAChRs and the brain’s response to it set the stage for receptor availability to become excessive. In male chronic smokers, nicotine occupies and desensitizes β2*-nAChRs in the cortex, striatum, and cerebellum. The brain compensates by adding new receptors. This adaptation restores roughly pre-smoking β2*-nAChR availability for as long as the man continues smoking. When he quits, however, the added receptors persist while nicotine leaves the brain, freeing up the β2*-nAChRs that it has been occupying. Together, the added and newly re-available β2*-nAChRs comprise an excess. In previous studies, the Yale researchers linked heightened β2*-nAChR availability to ex-smokers’ nicotine craving and possibly other withdrawal symptoms over 4 to 12 weeks of abstinence.
With sustained abstinence, the ex-smoker’s brain will eliminate the excess β2*-nAChRs. While excess β2*-nAChRs remain, however, NRT can occupy and desensitize some of them, just as nicotine from tobacco did, and so keep receptor availability at approximately pre-smoking levels. Used in individualized, tapering doses, NRT can thus suppress craving and other consequences of neuronal overstimulation while allowing the brain to gradually re-adapt to nicotine-free functioning.
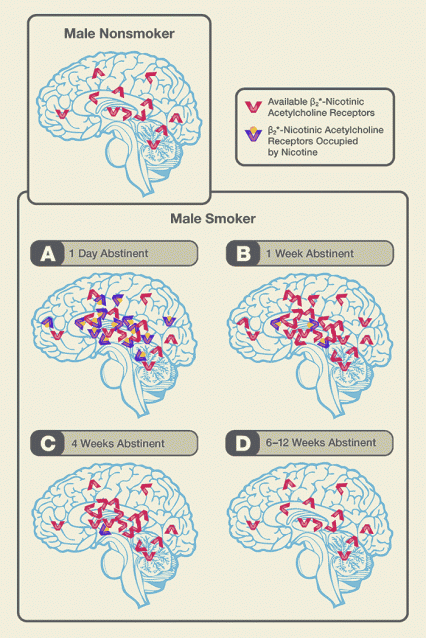 Figure 3. The Trajectory of β2*-nAChR Quantity and Availability During Smoking Abstinence in Males Heightened β2*-nAChR availability in a male ex-smoker's brain promotes nicotine craving. During the first 6-12 weeks of abstinence, β2*-nAChR availability follows a rising and falling trajectory determined by the rates at which a) nicotine leaves the brain and b) the brain clears receptors. Dr. Cosgrove and colleagues have shown that:
Figure 3. The Trajectory of β2*-nAChR Quantity and Availability During Smoking Abstinence in Males Heightened β2*-nAChR availability in a male ex-smoker's brain promotes nicotine craving. During the first 6-12 weeks of abstinence, β2*-nAChR availability follows a rising and falling trajectory determined by the rates at which a) nicotine leaves the brain and b) the brain clears receptors. Dr. Cosgrove and colleagues have shown that:A. On abstinence day 1, receptor quantity is elevated compared to levels seen in nonsmokers; availability is not markedly increased over that seen in nonsmokers.
B. At 1 week of abstinence, receptor quantity remains elevated; nicotine has vacated many of the receptors it previously occupied, and availability peaks.
C. At 4 weeks of abstinence, the brain has cleared a significant portion of the receptors generated during chronic smoking; nicotine has largely left the brain, and receptor availability is down from its peak.
D. After 6-12 weeks of abstinence, the brain has reduced receptor quantity to near pre-smoking levels; no nicotine remains, and availability is near nonsmokers' levels.
Women and Progesterone
“The study points to the importance of developing approaches to smoking cessation for women that do not rely on nicotine-containing treatments,” says Dr. Cosgrove. “Right now, if someone wants to quit smoking, everything over the counter and even prescribed drugs act on β2*-nAChRs in some way. For men, who have an upregulated pool of these receptors, these treatments can help wean them down. For women, who don’t appear to have β2*-nAChR upregulation, our findings suggest that we may need to target other receptors.”
The Yale results implicated progesterone as a potential source of difficulty for women who try to quit. Consistent with some other research, women with higher levels of progesterone reported more craving to smoke, worse withdrawal symptoms, and deeper depression than women with lower levels.
“To me, this finding suggests that the more progesterone you have, the worse you feel,” Dr. Cosgrove says. However, she notes that the role of progesterone in abstinence phenomena remains unclear. For example, in the Yale study:
- Higher progesterone blood levels corresponded both to reduced β2*-nAChR availability in all brain regions except the thalamus and to craving and withdrawal symptoms. These findings suggest that progesterone might promote women’s negative abstinence experiences by inhibiting β2*-nAChR availability. However, the study did not detect any direct link between women’s β2*-nAChR availability and craving or symptoms.
- Higher progesterone levels were significantly associated with craving and withdrawal symptoms, which suggests that lowering progesterone levels could help women quit smoking. However, some investigators have reported the opposite, that supplementing progesterone has helped women quit.
Further studies with different designs and participant populations will resolve these apparent conundrums, the researchers say.
Next Questions
“Our study makes sense of the findings that nicotine replacement therapies are not as effective in women and highlights that we need to look seriously at sex differences in all types of drug addiction,” Dr. Cosgrove says.
Dr. Mary Kautz of NIDA’s Division of Clinical Neuroscience and Behavioral Research says that Dr. Cosgrove’s findings are important because many of the most common treatments are nicotine-based. “This work shows that women may benefit more from non-nicotinic medications or behavioral therapies and relaxation techniques than they do from NRT. This is one of the first times we’ve seen evidence of a molecular mechanism that may underlie sex differences in success with smoking cessation.”
Dr. Kautz adds that the findings related to progesterone level “were a very interesting part of this study. Dr. Cosgrove will use them as a launching pad for other studies.”
Dr. Cosgrove is looking for other receptors to target as part of a newly funded center. In July 2012, her Yale University colleague Dr. Sherry McKee received a Specialized Center of Research (SCOR) grant from NIDA and the National Institutes of Health Office of Research on Women’s Health to develop effective smoking cessation treatments that account for gender differences such as those highlighted in the current study. Because women appear to smoke to even out emotions and regulate stress, the team will focus on the hypothalamic-pituitary-adrenal (HPA) axis, which regulates stress.
This study was supported by NIH grants: DA020651, DA20651, DA031750, DA21863, AA15632, DA13334, DA015577, and TR000142.
Source:
Cosgrove, K.P.; Esterlis, I.; McKee, S.A.; et al. Sex differences in availability of β2*-nicotinic acetylcholine receptors in recently abstinent tobacco smokers. Archives of General Psychiatry 69(4):418-427, 2012. Full Text
