This research:
- Identified hundreds of genes that promote or inhibit reactivation of latent HIV.
- Showed that inhibiting mTOR, a regulatory protein complex, can prevent reactivation of latent HIV.
- Suggested that medications to inhibit mTOR might help people with HIV achieve and maintain undetectable HIV viral loads.
Current treatment of human immunodeficiency virus (HIV) infection centers on the use of multiple medications that disrupt the virus’ lifecycle at several stages. The approach, called highly active antiretroviral therapy (HAART), halts progression of HIV disease, extends patients’ life expectancy and enables many to live relatively free of symptoms. However, HAART does not cure the disease, because it only targets virus that is actively replicating. People infected with HIV also carry a reservoir of inactive, or latent, virus that, under the right circumstances, can emerge from latency and reestablish disease progression.
The Verdin Laboratory at the Gladstone Institute of Virology and Immunology in San Francisco, working with an international team of researchers, applied cell- and molecular-biology approaches to broaden understanding of factors that affect reactivation of latent HIV. Such factors might provide targets for medications to permanently prevent disease progression, either by reactivating latent virus so that it becomes susceptible to HAART or by locking it in its latent state.
A Whole-Genome Screen
Dr. Eric Verdin and his team assessed how suppressing each gene in the human genome would affect reactivation of latent HIV. To accomplish this, they introduced a library of small molecules, called short hairpin RNA (shRNA), into engineered cells that were latently infected with the virus (see Figure 1). Each shRNA binds and inhibits one gene, and, collectively, the shRNA library inhibited every gene in the human genome. The researchers then exposed the cells to a pair of antibodies that stimulated reactivation of latent HIV in some, but not all, of the cells.
Fluorescent markers on the HIV DNA and shRNA enabled the researchers to compare gene expression in cells with shRNA-suppressed genes that did or did not show HIV reactivation. The comparison identified 335 genes that promote viral reactivation; cells in which these genes were suppressed exhibited less viral reactivation than cells in which these genes were active. Another 330 genes were found to support viral latency; cells in which these genes were suppressed showed increased reactivation.
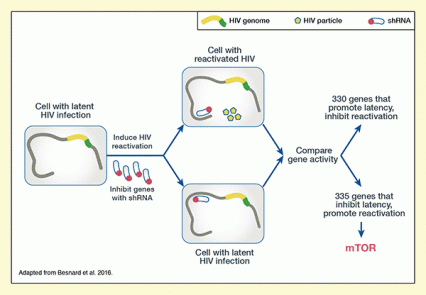 Figure 1. Identification of Genes that Regulate HIV Latency The researchers introduced gene-inhibiting short hairpin RNAs (shRNAs) into cells with latent HIV infection, then exposed the cells to antibodies (α-CD3/CD28) that trigger reactivation of latent HIV. Reactivation was increased in cells where shRNAs inhibited genes that promote HIV latency, and reduced in cells where shRNAs inhibited genes that promote reactivation. The researchers used an shRNA library that screened the entire genome, and identified 665 genes that affect HIV reactivation. They were especially intrigued to discover that genes related to the mTOR regulatory complex appear to inhibit activation of latent HIV.
Figure 1. Identification of Genes that Regulate HIV Latency The researchers introduced gene-inhibiting short hairpin RNAs (shRNAs) into cells with latent HIV infection, then exposed the cells to antibodies (α-CD3/CD28) that trigger reactivation of latent HIV. Reactivation was increased in cells where shRNAs inhibited genes that promote HIV latency, and reduced in cells where shRNAs inhibited genes that promote reactivation. The researchers used an shRNA library that screened the entire genome, and identified 665 genes that affect HIV reactivation. They were especially intrigued to discover that genes related to the mTOR regulatory complex appear to inhibit activation of latent HIV.
- Text description of Figure 1
-
The figure shows the experimental procedure used to identify genes promoting or inhibiting HIV reactivation. On the left is depicted a cell with latent HIV infection, indicated by the yellow/green line representing the HIV genome inserted into the cell’s DNA (brown line). These cells underwent treatment to induce HIV reactivation and gene inhibition with short hairpin RNA (shRNA) molecules, as indicated by a two-headed arrow pointing away from the cell. Oblong shapes with a red dot represent the shRNA molecules. As indicated by the two-headed arrow, this treatment resulted in two different cell populations: cells with reactivated HIV (top) and cells with latent HIV infection (bottom). In the cell with reactivated HIV, an shRNA is located next to one end of the DNA, indicating that it has bound to a gene there. The cell also contains HIV particles represented by the yellow pentagons. In the cell with the latent HIV infection, the shRNA is located next to another area of the DNA, indicating that it has bound to a different gene. This cell has no HIV particles. Two arrows pointing towards each other from these cells indicate that the researchers compared the gene activity of the two cell populations. Two arrows pointing away from each other on the right side indicate that the analyses identified 2 groups of genes: one group of 330 genes promoted latency and inhibited HIV reactivation; another group of 335 genes inhibited latency and promoted reactivation. An arrow points down from this group towards the red word “mTOR” to indicate that mTOR was identified as one of these genes.
From Gene to Protein Complex
Several of the genes identified by the genetic screen encode proteins that were already known to affect HIV latency and reactivation. The researchers took particular note of a set of genes that encode proteins for an important regulatory complex not previously implicated in HIV latency and reactivation.
The complex, called mTOR (mammalian target of rapamycin), helps coordinate cellular growth in response to environmental or cellular influences. Two variants, mTORC1 and mTORC2, regulate biological processes ranging from lipid metabolism to cell proliferation. In addition to the current study, some of what is known about mTOR suggests that the complex might promote HIV reactivation. For example, mTOR interacts with other regulatory pathways that have been shown to affect HIV latency and reactivation, including transforming growth factor beta (TGF-β) signaling and actin remodeling.
A Potential Treatment?
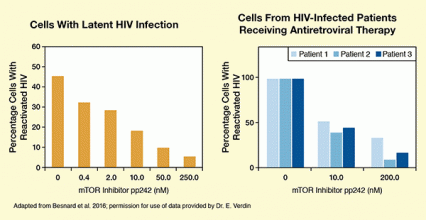 Figure 2. Inhibition of mTOR Prevents Reactivation of Latent HIV in Cultured Cells and Cells From HIV-Positive Patients (left) Cells with latent HIV infection were treated with antibodies (α-CD3/CD28) to induce HIV reactivation as described in Figure 1. The antibodies triggered viral reactivation in about 45 percent of the cells. Treating the cells simultaneously with the antibodies and the mTOR inhibitor pp242 reduced the percentage of cells with HIV reactivation, with higher doses producing greater reductions. (right) α-CD3/CD28, triggered reactivation of HIV in 100 percent of cells from isolated from three HIV-infected patients who were on antiretroviral therapy and had an undetectable viral load (i.e., had latent HIV infection). Treatment with increasing amounts of the mTOR inhibitor pp242 progressively reduced the percentage of reactivated cells, again confirming that mTOR promotes HIV reactivation.
Figure 2. Inhibition of mTOR Prevents Reactivation of Latent HIV in Cultured Cells and Cells From HIV-Positive Patients (left) Cells with latent HIV infection were treated with antibodies (α-CD3/CD28) to induce HIV reactivation as described in Figure 1. The antibodies triggered viral reactivation in about 45 percent of the cells. Treating the cells simultaneously with the antibodies and the mTOR inhibitor pp242 reduced the percentage of cells with HIV reactivation, with higher doses producing greater reductions. (right) α-CD3/CD28, triggered reactivation of HIV in 100 percent of cells from isolated from three HIV-infected patients who were on antiretroviral therapy and had an undetectable viral load (i.e., had latent HIV infection). Treatment with increasing amounts of the mTOR inhibitor pp242 progressively reduced the percentage of reactivated cells, again confirming that mTOR promotes HIV reactivation.
- Text description of Figure 2
-
The two bar charts show the percentage of cells with reactivated HIV among either cultured cells with latent HIV infection (left panel) or cells from HIV-infected patients receiving antiretroviral therapy (right panel). In the left panel, the cells were treated with increasing concentrations of the mTOR inhibitor pp242. In the absence of pp242, about 45 percent of cells showed HIV reactivation. With 0.4 nM pp242, the percentage of cells with reactivated HIV was about 32 percent; with 2.0 nM pp242 it was about 28 percent; with 10.0 nM pp242 it was about 18 percent; with 50.0 nM pp242 it was about 10 percent; and with 250.0 nM pp242 it was about 5 percent. The right panel shows the results for cells isolated from three different HIV-infected patients, indicated by different shades of blue. The first set of bars shows the percentage of cells with reactivated HIV after HIV reactivation but in the absence of pp242. Under these conditions, 100 percent of cells from all three patients showed reactivation. The second set of bars shows the percentage of cells with reactivated HIV after HIV reactivation and treatment with 10.0 nM pp242. Under these conditions, about 50 percent, 40 percent, and 45 percent or cells, respectively, showed HIV reactivation. The third set of bars shows the percentage of cells with reactivated HIV after HIV reactivation and treatment with 200.0 nM pp242. Under these conditions, about 35 percent, 10 percent, and 20 percent of cells, respectively, showed HIV reactivation.
The Verdin Lab researchers reasoned that if mTOR plays a key role in reactivating latent HIV, inhibiting the complex might lock the virus in its latent state. In a new experiment using a different engineered cell line, the researchers demonstrated that, indeed, debilitating the complex (by knocking down its subunit proteins) halted HIV reactivation. Importantly, they duplicated this finding in CD4 cells from patients infected with HIV (see Figure 2).
“The identification of the mTOR pathway as a key regulator of latency and reactivation opens up new therapeutic approaches for preventing HIV from reactivating,” says Dr. Verdin. He notes that several mTOR inhibitors are under development and being tested as cancer therapeutics. Future research will evaluate the effect of these mTOR inhibitors on HIV reservoir suppression and identify candidate targets in the mTOR signaling pathway.
Dr. Amy Lossie, a Program Officer in the Genetics, Epigenetics, and Developmental Neuroscience Branch at NIDA, says that certain patient groups in particular could benefit if mTOR blockade to prevent HIV reactivation proves clinically viable. Dr. Lossie explains, “Some patients who are on HAART continue to have detectable virus in their blood, for example, due to low medication adherence. Adding mTOR inhibitors to HAART could help these patients achieve undetectable viral loads.”
Preventing HIV reactivation is an alternative to another approach that researchers are pursuing as a potential answer to the problem of latent HIV. That approach, called “shock-and-kill,” reactivates latent virus, rendering it susceptible to HAART medications. In theory, if it were possible to shock-and-kill all the latent virus in a patient’s body, patients could be cured of HIV disease. However, Dr. Lossie says, “There are hidden reservoirs of HIV, such as in glia cells in the brain, that are particularly hard to kill with this approach.” Although the alternative approach will not cure the disease, medications that successfully lock HIV in its reservoirs would enhance the efficacy of HAART and might even be sufficient therapy in themselves for patients who have achieved complete viral suppression.
This study was supported by NIH grant DA041742.
Source
Besnard, E.; Hakre, S.; Kampmann, M.; et al. The mTOR complex controls HIV latency. Cell Host Microbe 20(9):785-797, 2016.
