This study found that:

- Memories for cocaine use weaken briefly after retrieval following prolonged abstinence.
- This instability in the strength of cocaine memories results from temporary changes in specific synaptic connections in the nucleus accumbens.
- Preventing cocaine memories from re-strengthening in the unstable period following retrieval attenuates cocaine seeking.
Many people who use cocaine and other drugs return to drug use even after prolonged abstinence. Resumption of drug use often is driven by drug-associated memories that are retrieved when the person is exposed to drug cues. The results of a recent study in rats suggest that some neuronal connections (i.e., synapses) that encode cocaine-associated memories are not static but weaken for approximately 6 hours after they are retrieved and that holding these synapses in their weakened state may reduce return to cocaine use.
Cocaine Use Generates New Synapses in the Nucleus Accumbens
The study presents the latest findings of a long-term project spearheaded by Dr. Yan Dong at the University of Pittsburgh that is aimed at understanding how the activity of specific synapses in the brain’s reward circuit mediates drug seeking and return to cocaine use. This research previously determined that in rats, cocaine experience generates new synaptic connections between certain neurons of the nucleus accumbens (NAc).
The synapses of interest are known as “silent synapses”: Although they respond to the neurotransmitter glutamate, they initially lack a specific type of glutamate receptor called the AMPA receptor (AMPAR) and therefore are considered AMPAR silent. Subsequent research by Dr. Dong and his team showed that over time (e.g., during drug withdrawal), these newly formed synapses “mature” by recruiting AMPARs and thereby strengthen. Researchers hypothesize that these synapses encode drug-associated memories and help sustain drug-seeking behavior.
Silent Synapses Are Unstable After Memory Retrieval and Can Be Disrupted
In the most recent study, Dr. William J. Wright, Dr. Nicholas M. Graziane, and Dr. Dong from the University of Pittsburgh and Penn State College of Medicine, along with colleagues from other institutions, examined what happens with those silent synapses when cocaine-associated memories are retrieved. Using a rat model of cocaine exposure, memory retrieval, and cue-induced cocaine seeking, the investigators assessed the excitability, morphology, and biochemical signaling of NAc neurons and their silent synapses as well as their effects on cue-induced cocaine seeking.
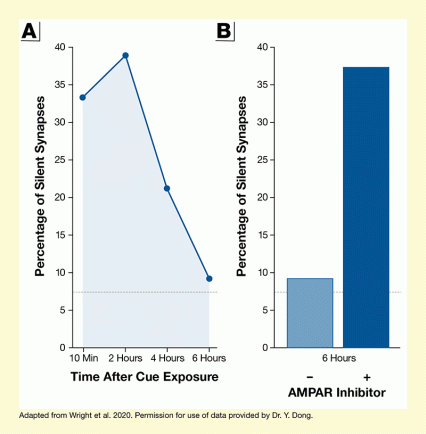
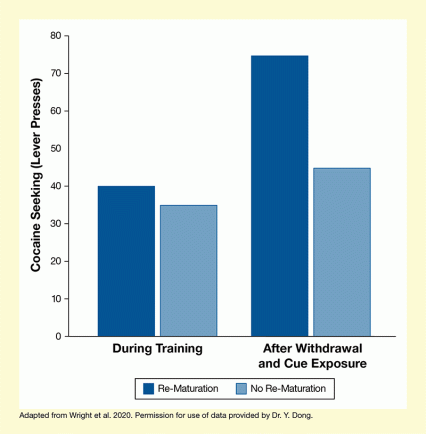
The researchers trained rats to associate a cue (e.g., a flash of light) with the availability of cocaine delivered after a lever press. After this association was established, the animals underwent prolonged cocaine abstinence, allowing the silent synapses to mature. Next, the researchers presented the cue again (i.e., the rats retrieved the cue-associated memory), triggering drug seeking in the animals. When the team looked at the NAc synapses after the cue exposure, they found that the matured synapses became AMPAR silent again for 6 hours, before re-maturing (see Figure 1). This re-maturation could be prevented by treating the animals with a compound that blocks AMPARs from being recruited (i.e., keeps the synapses silent). More importantly, additional experiments found that when the investigators blocked the re-maturation of silent synapses, the rats’ cocaine seeking was significantly reduced (see Figure 2).
The researchers speculate that when the silent synapses are prevented from re-stabilizing following cue-associated memory retrieval, at least some of the animals’ cocaine-associated memories are disrupted, which may reduce their urge to seek out more of the drug. According to Dr. Dong, “These findings have broader implications for the field in that they provide concrete cellular substrates that mediate the dynamics of drug memories. As such, a cellular determinant of drug memories is characterized.”
This work was supported by NIDA grants DA043940, DA023206, DA044538, DA040620, DA047861, DA035805, DA008227, and DA014133 as well as the NIDA Intramural Research Program.
- Text Description of Figure 1
-
The figure illustrates the changes in the percentage of silent synapses in rats after long-term withdrawal from cocaine and re-exposure to cocaine-associated cues. The left panel plots the percentage of silent synapses against time after cue exposure. The horizontal x-axis shows time after cue exposure from 10 minutes to 6 hours. The vertical y-axis shows the percentage of silent synapses on a scale from 0 to 40. The percentage of silent synapses was about 33 percent at 10 minutes, about 39 percent at 2 hours, about 22 percent at 4 hours, and about 9 percent at 6 hours.
The right panel shows the percentage of silent synapses at 6 hours after cue exposure in the absence or presence of an AMPAR inhibitor. The vertical y-axis again shows the percentage of silent synapses on a scale from 0 to 40. The left, lighter blue bar represents synapses in the absence of the AMPAR inhibitor; the percentage of silent synapses was about 9 percent. The right, dark blue bar represents synapses in the presence of the AMPAR inhibitor; the percentage of silent synapses was about 37 percent, indicating prevention of re-maturation of the silent synapses.
- Text Description of Figure 2
-
The bar chart shows the effect of preventing re-maturation of silent synapses on cocaine seeking. Dark blue bars represent animals in which re-maturation of silent synapses occurred; lighter blue bars represent animals in which re-maturation of silent synapses was prevented. The left pair of bars represents animals tested during training, and the right pair of bars represents animals tested after withdrawal and re-exposure to cocaine cues. The vertical y-axis shows cocaine seeking as indicated by number of lever presses on a scale from 0 to 80.
In the animals tested during training, those in which re-maturation of silent synapses occurred performed about 40 lever presses and those in which re-maturation was prevented performed about 35 lever presses. In the animals that were tested after withdrawal and cue exposure, those with re-maturation of silent synapses performed about 75 lever presses and those with no re-maturation of silent synapses performed about 45 lever presses.
Source:
- Wright, W.J., Graziane, N.M., Neumann, P.A., et al. Silent synapses dictate cocaine memory destabilization and reconsolidation. Nat Neurosci 23(1):32-46, 2020.