This research:
- Sheds light on a key mechanism of adolescent brain development.
- Shows how amphetamine disrupts this mechanism.
- Links this disruption to animal behaviors that resemble human susceptibility to addiction.
During adolescence, the prefrontal cortex undergoes elaboration and refinement of the neural circuits that underpin adult self-control and mature judgment. Dr. Cecilia Flores’ team, with colleagues from McGill and Concordia Universities in Montreal, conducted experiments with mice to understand this process and how drugs affect it.
The researchers found that some dopamine axons that reach the midbrain early in life extend into the prefrontal cortex (PFC) during adolescence. Their experiments indicate that a protein called DCC controls this process. DCC resides in the tips of growing dopamine axons and uses molecular signals emitted by neurons as guideposts to pilot the axons to target sites. Upon reaching their target sites, the axons form synapses with neighboring neurons.
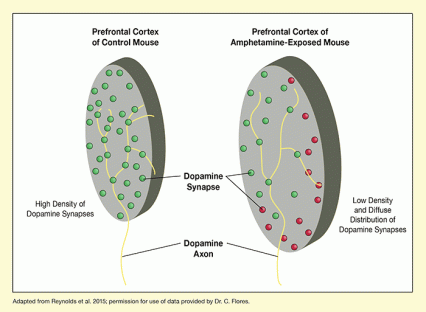
The researchers demonstrated that amphetamine alters the course of axon growth and synapse formation in adolescence. In these experiments, they exposed adolescent mice to amphetamine in amounts similar to those used in human amphetamine abuse, and examined the animals’ brain tissue at full maturity. Dopamine axons in the PFC of the exposed mice were spread out more diffusely and formed fewer synapses than axons in unexposed mice (see Figure 1). In behavioral tests, the mice exposed to amphetamine in adolescence exhibited enhanced sensitivity to the drug’s rewarding effects and potential for abuse.
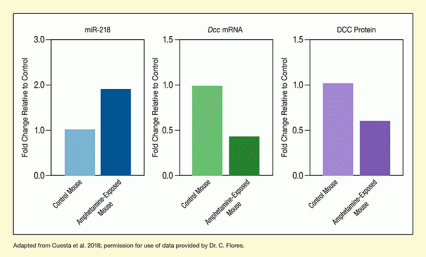
Using a variety of genetic and molecular techniques, the Canadian researchers showed that amphetamine disrupts normal development and synapse formation of dopamine neurons by suppressing DCC production. The drug causes a very short strip of RNA (miR-218) to attach to Dcc messenger RNA and inhibit its translation into protein (see Figure 2). With less DCC to guide axons and facilitate synapse formation, the final organization and connectivity of the PFC are compromised.
The consequences of amphetamine-altered adolescent PFC development include deficits in cognition and heightened susceptibility to addiction phenotypes in adulthood. Dr. Flores suggests that because the causal sequence her group uncovered links alterations in miR-218 to susceptibility to these effects, it may be possible to use miR-218 levels to predict risk. She says, “We are now interested in determining whether changes in levels of miR-218 in specific brain regions can be detected in blood so as to be able to ask whether circulating levels of this microRNA in adolescence can predict differential vulnerability to enduring effects of drugs of abuse or stressors.”
Dr. Flores stresses that the amphetamine doses that altered brain development in this study were high, equivalent to those seen in illicit drug use. She and her colleagues are currently assessing whether low doses, such as those used to treat attention-deficit/hyperactivity disorder, might produce similar effects. Their preliminary results suggest that they do not.
This research was supported by NIH grant DA037911.
- Text Description of Figure 1
-
The figure shows graphic representations of tissue sections from the prefrontal cortex of a control mouse (left) and an amphetamine-exposed mouse (right). The yellow lines entering the tissue sections from the bottom represent dopamine axons that split and form synapses in the prefrontal cortex. Green circles represent the normal synapses that are closely spaced in the prefrontal cortex of the control mouse. Some of these synapses can also be found in the prefrontal cortex of the amphetamine-exposed mouse (green circles), but at a lower density. Also, additional dopamine synapses are spread out beyond the normal area in the amphetamine-exposed mouse, indicated by red circles.
- Text Description of Figure 2
-
The three bar charts show the differences in the levels of miR-218, Dcc mRNA, and DCC protein between control mice (lighter colors) and amphetamine-exposed mice (darker colors). In all three charts, the level of the variable measured is set as 1.0 for the control mouse and the level for the amphetamine-exposed mouse is shown as the fold change relative to the control mouse. The left chart (blue bars) illustrates the levels of miR-218. The level is 1.0 for the control mouse and approximately 1.9 for the amphetamine-exposed mouse. The middle chart (green bars) shows the levels of Dcc mRNA. The level is again 1.0 for the control mouse and approximately 0.4 for the amphetamine-exposed mouse. The right chart (purple bars) shows the levels of DCC protein. The level is again set at 1.0 for the control mouse and is approximately 0.6 for the amphetamine-exposed mouse.
Sources:
- Cuesta, S., Restrepo-Lozano, L.M., Silvestrin, S., et al. Non-contingent exposure to amphetamine in adolescence recruits miR-218 to regulate Dcc expression in the VTA. Neuropsychopharmacology 43(4):900-911, 2018.
- Reynolds, L.M., Makowski, C.S., Yogendran, S.V., et al. Amphetamine in adolescence disrupts the development of medial prefrontal cortex dopamine connectivity in a DCC-dependent manner. Neuropsychopharmacology 40(5):1101-12, 2015.
- Reynolds, L.M., Pokinko, M., Torres-Barrío, A., et al. DCC Receptors Drive Prefrontal Cortex Maturation by Determining Dopamine Axon Targeting in Adolescence. Biol Psychiatry 83(2):181-192, 2018.